Frontiers | Mu-opioid receptor and receptor tyrosine kinase crosstalk: Implications in mechanisms of opioid tolerance, reduced analgesia to neuropathic pain, dependence, and reward

An Opioid Agonist that Does Not Induce μ-Opioid Receptor—Arrestin Interactions or Receptor Internalization | Molecular Pharmacology
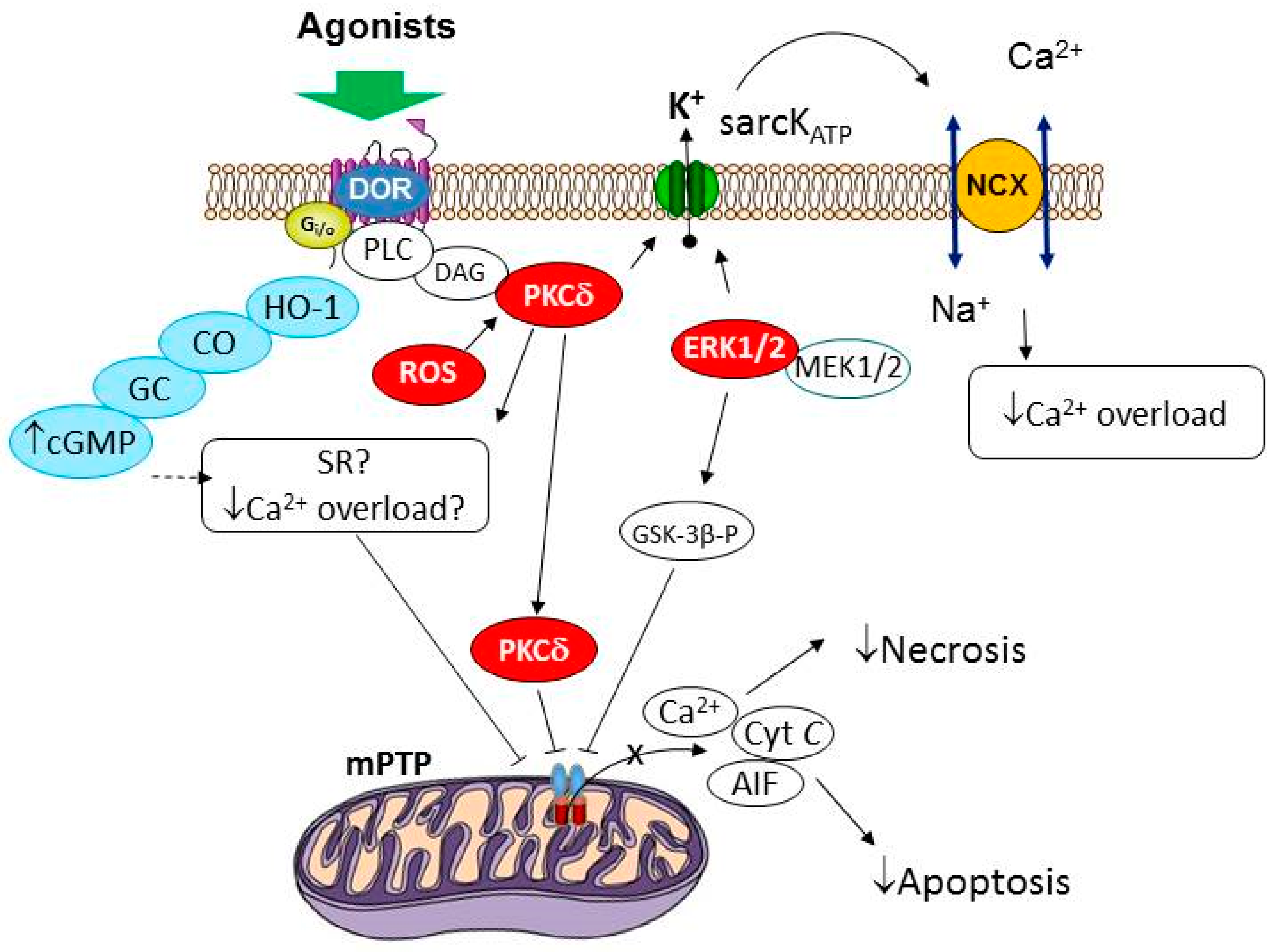
Biomedicines | Free Full-Text | δ-Opioid Receptor as a Molecular Target for Increasing Cardiac Resistance to Reperfusion in Drug Development

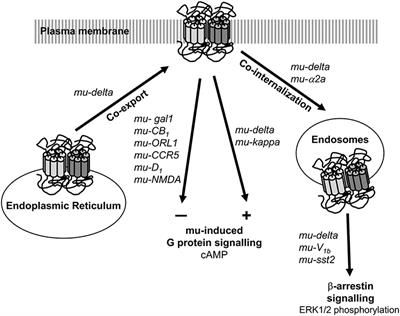
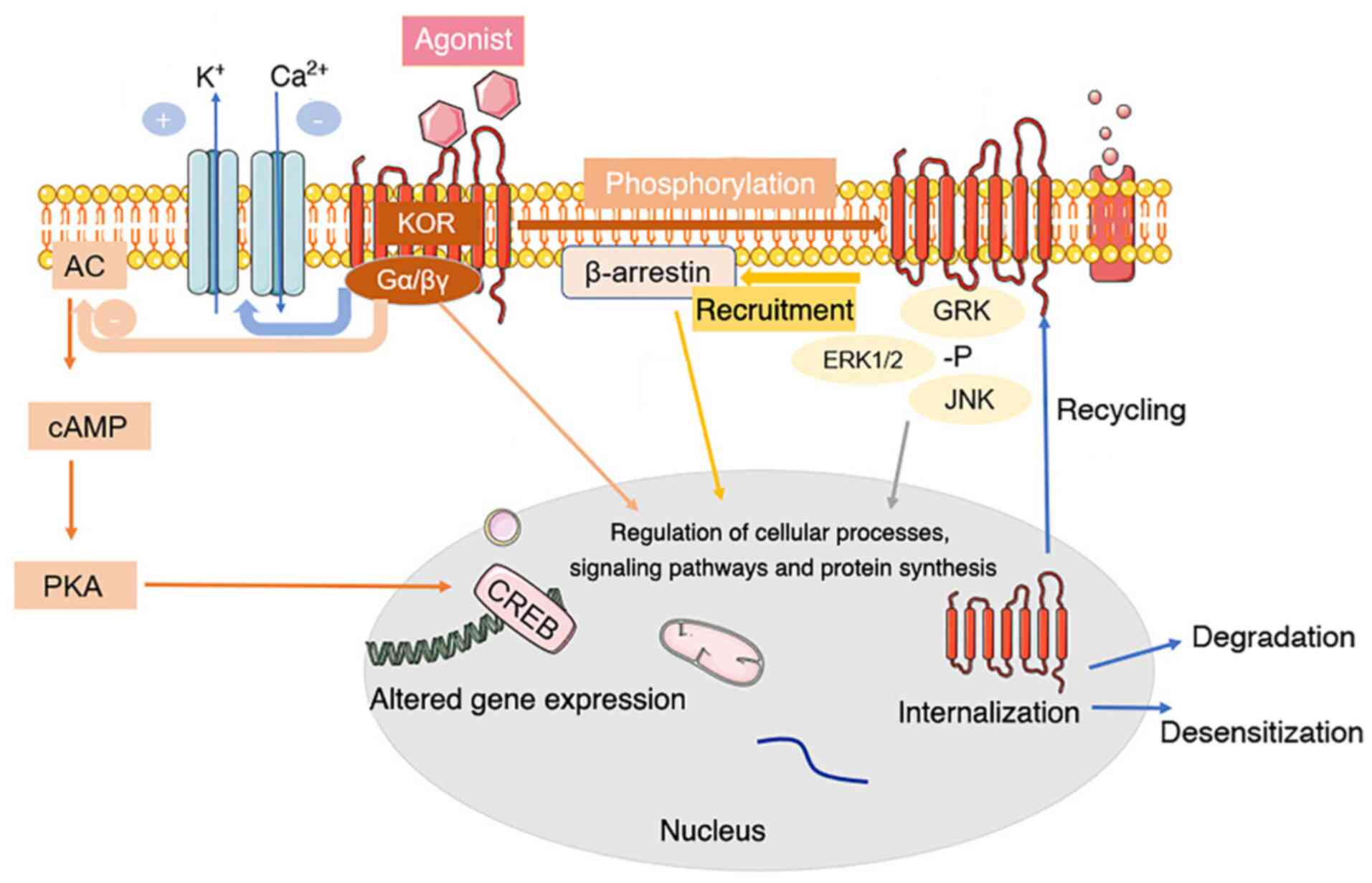
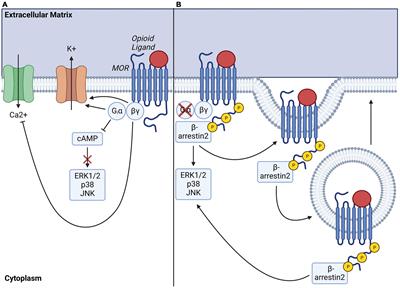
Regulation of µ-Opioid Receptors: Desensitization, Phosphorylation, Internalization, and Tolerance | Pharmacological Reviews
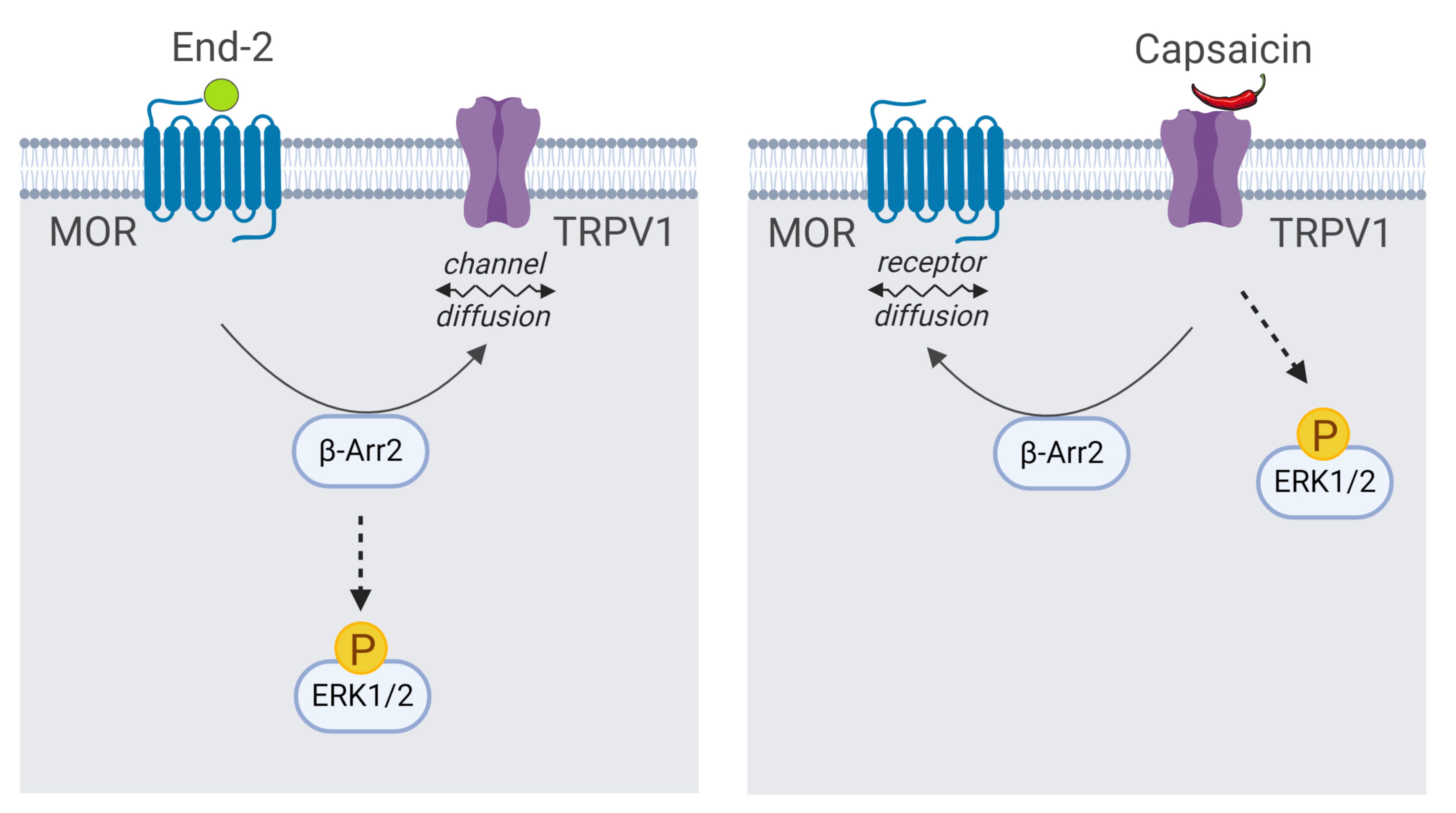
IJMS | Free Full-Text | β-Arrestin 2 and ERK1/2 Are Important Mediators Engaged in Close Cooperation between TRPV1 and µ-Opioid Receptors in the Plasma Membrane
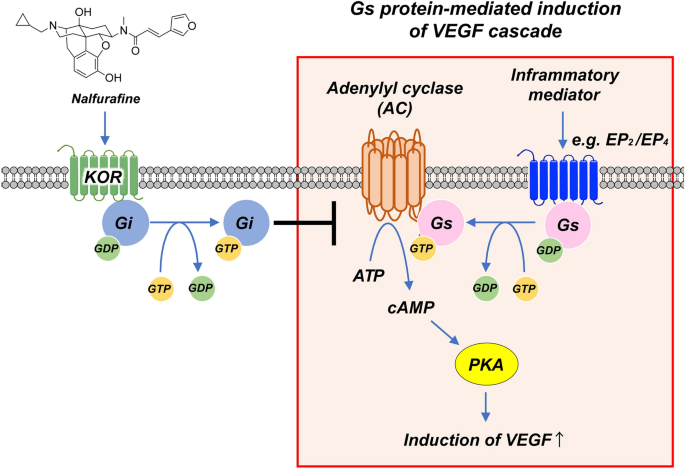
Topical administration of the kappa opioid receptor agonist nalfurafine suppresses corneal neovascularization and inflammation | Scientific Reports

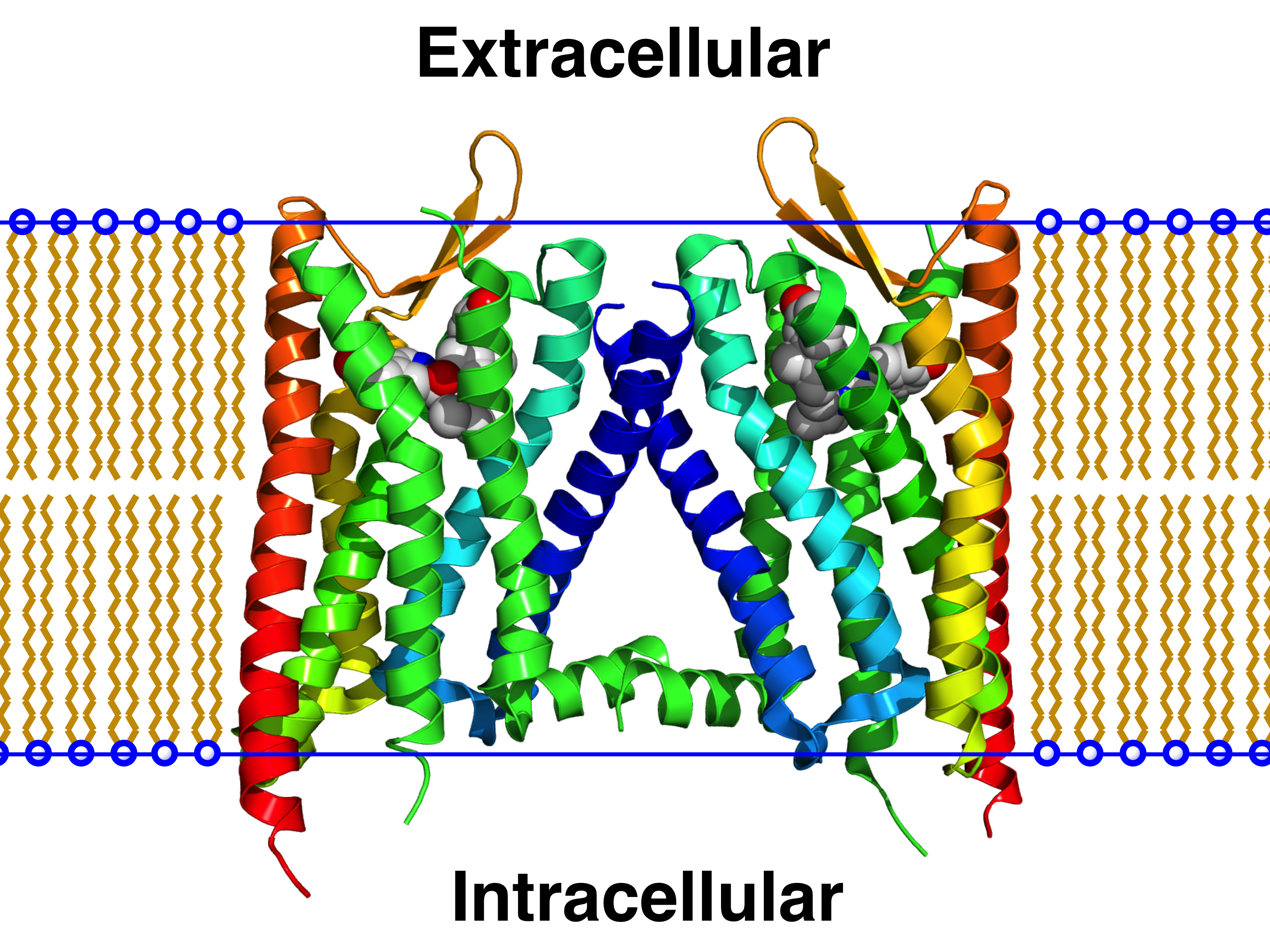
Signaling Properties of Structurally Diverse Kappa Opioid Receptor Ligands: Toward in Vitro Models of in Vivo Responses | ACS Chemical Neuroscience

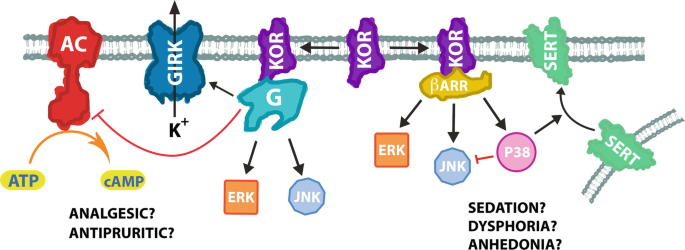
Biased agonists of the kappa opioid receptor suppress pain and itch without causing sedation or dysphoria | Science Signaling

Constitutive activation of kappa opioid receptors at ventral tegmental area inhibitory synapses following acute stress | eLife

Mitogen‐activated protein kinase signaling mediates opioid‐induced presynaptic NMDA receptor activation and analgesic tolerance - Deng - 2019 - Journal of Neurochemistry - Wiley Online Library
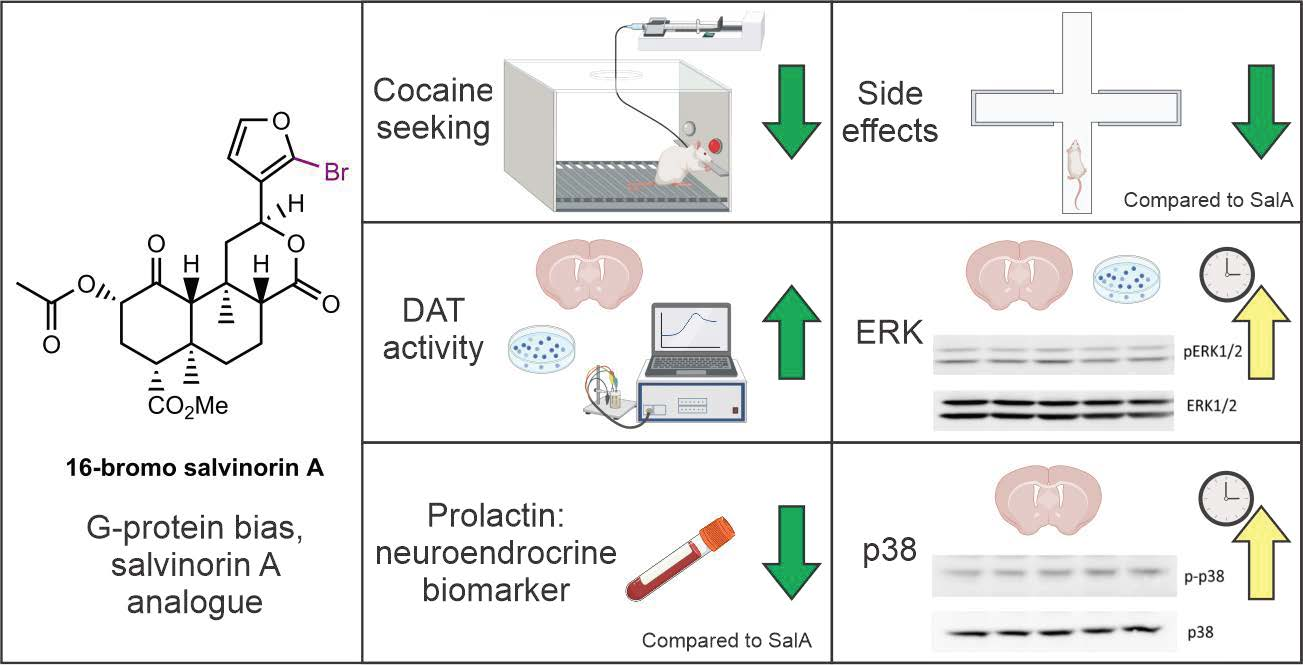
Molecules | Free Full-Text | The Kappa Opioid Receptor Agonist 16-Bromo Salvinorin A Has Anti-Cocaine Effects without Significant Effects on Locomotion, Food Reward, Learning and Memory, or Anxiety and Depressive-like Behaviors

Nalfurafine is a G-protein biased agonist having significantly greater bias at the human than rodent form of the kappa opioid receptor - ScienceDirect

β-Arrestin-Dependent μ-Opioid Receptor-Activated Extracellular Signal-Regulated Kinases (ERKs) Translocate to Nucleus in Contrast to G Protein-Dependent ERK Activation | Molecular Pharmacology
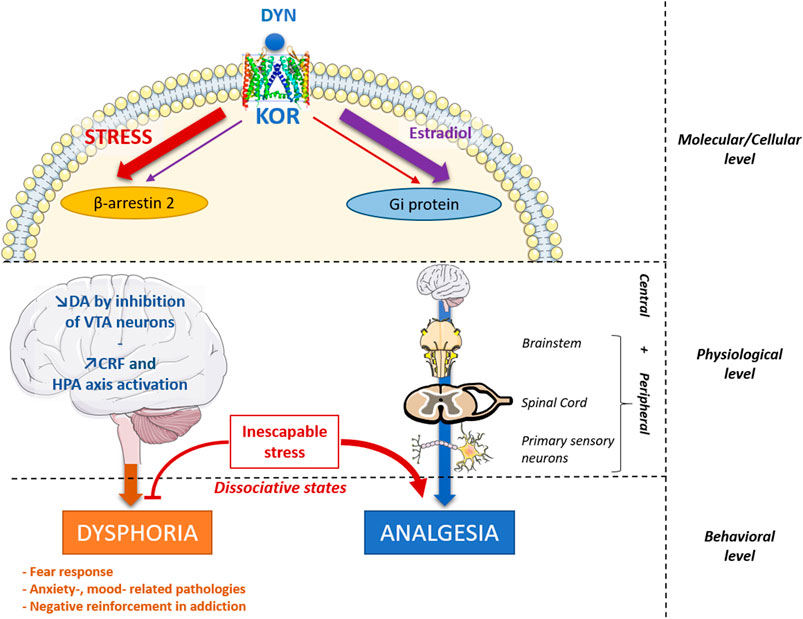
Frontiers | Traumatic Stress-Induced Vulnerability to Addiction: Critical Role of the Dynorphin/Kappa Opioid Receptor System
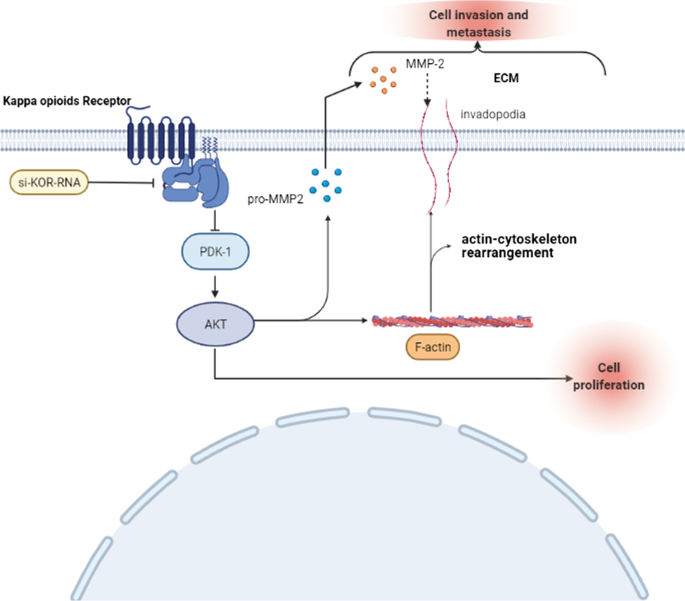
RETRACTED ARTICLE: Down-regulation of kappa opioid receptor promotes ESCC proliferation, invasion and metastasis via the PDK1-AKT signaling pathway | Cell Communication and Signaling | Full Text